Styrene-butadiene rubber 1502
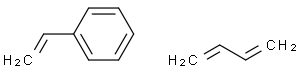
Styrene, 1,3-butadiene polymer
CAS: 9003-55-8
Molecular Formula: C12H14
Styrene-butadiene rubber 1502 - Names and Identifiers
Styrene-butadiene rubber 1502 - Physico-chemical Properties
| Molecular Formula | C12H14 |
| Molar Mass | 158.24 |
| Density | 1.04 g/mL at 25 °C |
| Melting Point | -59 |
| Appearance | Morphology slab/chunk |
| PH | 10~13 |
| Storage Condition | Room Temprature |
| Stability | Stable. Combustible. Incompatible with strong oxidizing agents. |
| Refractive Index | 1.57 |
| Physical and Chemical Properties | The chemical properties include liquid latex (such as American SBR2000 type) and solid rubber (such as American SBRl028 type). Styrene odor, not completely soluble in gasoline, benzene and chloroform. The relative density is 0.9~0.95, and the glass transition temperature is -60 ℃ ~-75 ℃. The pH value of 50/50 latex is 10.0~11.5, and the solid content is 41% ~ 63%. The pH value of 75/25 latex is 9.5~11.0, and the solid content is 26% ~ 42%. |
| Use | Mainly used in the manufacture of tires, transport belts, rubber tubes, adhesive sponge rubber, impregnated fibers and fabrics, can also be directly used as adhesives, coatings, etc |
Styrene-butadiene rubber 1502 - Risk and Safety
| WGK Germany | 3 |
| RTECS | WL6478000 |
Styrene-butadiene rubber 1502 - Upstream Downstream Industry
Styrene-butadiene rubber 1502 - Nature
Open Data Verified Data
There are two forms of liquid latex and fixed rubber. Styrene odor, not completely soluble in gasoline, benzene and chloroform.
Last Update:2024-01-02 23:10:35
Styrene-butadiene rubber 1502 - Preparation Method
Open Data Verified Data
latex was prepared by emulsion copolymerization of butadiene and styrene with rosin acid soap or fatty acid soap as emulsifier and persulfate as initiator, and then coagulated and dried to obtain solid rubber. The 75/25 solid rubber can also be prepared by solution copolymerization of butadiene and benzene Diene in hexane with butyl lithium as catalyst.
Last Update:2022-01-01 10:08:55
Supplier List
Featured ProductsSpot supply
Product Name: Polystyrene-block-polybutadiene-block-polystyrene styrene 30 wt. % Visit Supplier Webpage Request for quotationCAS: 9003-55-8
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Benzene, ethenyl-, polymer with 1,3-butadiene, hydrogenated Request for quotation
CAS: 9003-55-8
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 9003-55-8
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Product Name: Styrene, 1,3-butadiene polymer Request for quotation
CAS: 9003-55-8
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
CAS: 9003-55-8
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
Product Name: Sbs Mixture Visit Supplier Webpage Request for quotation
CAS: 9003-55-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 9003-55-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Featured ProductsSpot supply
Product Name: Polystyrene-block-polybutadiene-block-polystyrene styrene 30 wt. % Visit Supplier Webpage Request for quotationCAS: 9003-55-8
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Benzene, ethenyl-, polymer with 1,3-butadiene, hydrogenated Request for quotation
CAS: 9003-55-8
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 9003-55-8
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Product Name: Styrene, 1,3-butadiene polymer Request for quotation
CAS: 9003-55-8
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
CAS: 9003-55-8
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
Product Name: Sbs Mixture Visit Supplier Webpage Request for quotation
CAS: 9003-55-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 9003-55-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History
Raw Materials for Styrene-butadiene rubber 1502
Downstream Products for Styrene-butadiene rubber 1502
Neoprene adhesive
TIRE
emulsion waterproof agent
styrene-butadiene block copolymer shape memory materials
Adhesive for plastic film 402
TIRE
emulsion waterproof agent
styrene-butadiene block copolymer shape memory materials
Adhesive for plastic film 402



